For example, the annihilation of an electron and an anti electron(positron) produces two photons: No reaction has ever been found that creates or destroyscharge. The experiments strongly suggested that chargeis quantized.Īnother important property of charge is that charge a conservedquantity. For example, he observed droplets with a charge equal to+/- e, +/- 2 e, +/- 3 e, etc., but never droplets with a charge equal to +/-1.45 e, +/- 2.28 e, etc. Millikan discovered that the charge on the oildroplets was always a multiple of the charge of the electron (e, thefundamental charge). Charge Quantization and Charge ConservationĪn important experiment in which the charge of small oil droplets wasdetermined was carried out by Millikan (details of this experiment will bediscussed in Chapter 23). The gravitational (F g) and Coulomb(F c) between the building blocks of atoms.Ģ2.2. The combined electrostatic andmagnetic force is called the electromagnetic force. If thecharges are moving with a uniform velocity, they will experience both theelectrostatic force and a magnetic force. The electric forceexerted under these circumstances is called the electrostatic force. Our discussion of the electric force will initially concentrate on those casesin which the charges are at rest or are moving very slowly. The attractive force between the electrons in one body and theprotons in the other body is exactly canceled by the repulsive force betweenthe electrons in the two bodies. Since mostmacroscopic objects are neutral, they have an equal number of protons andelectrons. This table showsclearly that the electric force dominates the motion of electrons in atoms.However, on a macroscopic scale, the gravitational force dominates. Table 22.3lists the gravitational and the Coulomb force between electrons, protons andneutrons when they are separated by 1 x 10 -10 m. Electric force between two charged objects.Īn important difference between the electric force and thegravitational force is that the gravitational force is always attractive, whilethe electric force can be repulsive (F c > 0), zero, or attractive(F c < 0), depending on the charges of the particles. This formula applies to elementary particles and small charged objects as longas their sizes are much less than the distance between them.įigure 22.1. 0 is he permittivity constant: 0 =8.85 x 10 -12 C 2/(N. R is the distance between particle 1 and particle 2 (see Figure 22.1) Q 1 and q 2 are the charges of particle 1 and particle 2,respectively Electric charges of the building blocks of atoms The direction of theforce is along the line joining the particles. " The magnitude of the electric force that a particle exerts on anotherparticle is directly proportional to the product of their charges and inverselyproportional to the square of the distance between them. The precise magnitude of the electric force that a charged particle exerts onanother is given by Coulomb's law: Since atoms are neutral, the number of electronsmust be equal to the number of protons. Detailed measurements have shown thatthe magnitude of the charge of the proton is exactly equal to the magnitude ofthe charge of the electron. The electric charge of electrons, protonsand neutrons are listed in Table 22.2. Per definition, the electric charge on a glassrod rubbed with silk is positive. The electric charge can benegative, zero, or positive. The unit ofelectric charge q is the Coulomb (C). Instead,it depends on a new quantity: the electric charge. 
This impliesthat the electric force does not depend on the mass of the particle. The attractive force between the electronsand the nucleus is called the electric force.Įxperiments have shown that the electric force between two objects isproportional to the inverse square of the distance between the two objects.The electric force between two electrons is the same as the electric forcebetween two protons when they are placed as the same distance. Measurements of the velocity of theorbital electrons in an atom have shown that the attractive force between theelectrons and the nucleus is significantly stronger than the gravitationalforce between these two objects. 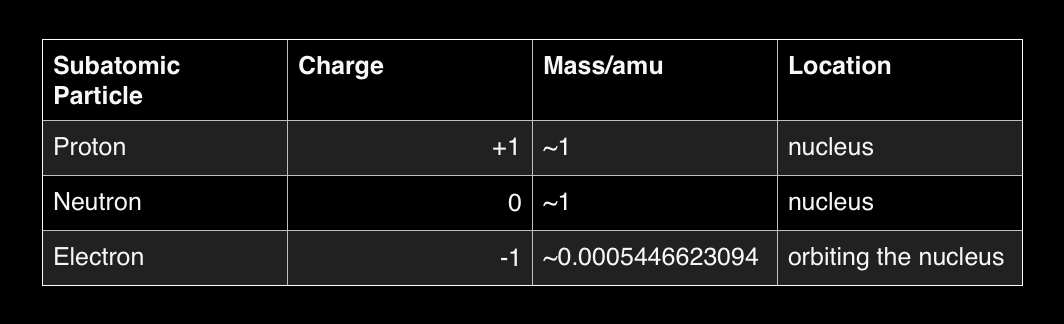
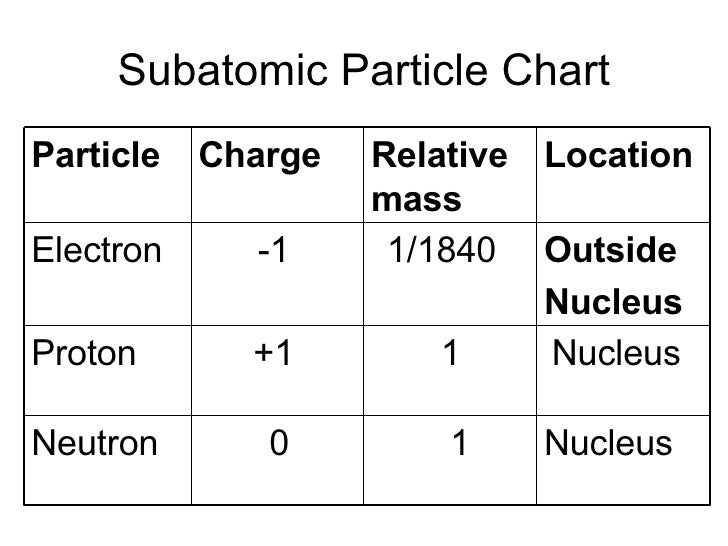
In P121 it was shown that anobject can only carry out circular motion if a radial force (directed towardsthe center of the circle) is present. The electrons are contained in a roughly spherical regionwith a diameter of about 2 x 10 -10 m. The diameter of the nucleus is between 10 -15 and10 -14 m. Mostof the mass of the atom is due to the mass of the nucleus. Themasses of the electrons, protons and neutrons are listed in Table 22.1. Each atom consists of a nucleus,consisting of protons and neutrons, surrounded by a number of electrons. Charge Quantization and Charge Conservation ELECTRIC FORCE AND ELECTRIC CHARGE CHAPTER 22 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
